COMPARISON OF TWO VALIDATED METHODS FOR CRYOPRESERVING EUROPEAN EEL (A. anguilla) SPERM: STANDARDIZATION AS A TARGET
Introduction
During the last years our groups have been working in parallel to develop cryopreservation protocols of European eel sperm. Each group established its own protocol in Spain (Asturiano et al., 2003; Peñaranda et al., 2009) and Hungary (Müller et al., 2004; Szabó et al., 2005) but they are different in many aspects (cryoprotectants, volumes, freezing media, grade for dilution, etc.) evidencing the need of standardization. Recent experiments proved the efficacy of the "Spanish method" by getting larvae of this species using frozen-thawed sperm (Asturiano et al., 2013). On the other hand, the Hungarian team was able to generate hybrid larvae using frozen-thawed sperm of European eel and Japanese eel eggs (Müller et al., 2012), which also indicates the validity of the "Hungarian method".
In the framework of the AQUAGAMETE COST Action, a pilot joint experiment was done in order to standardize the eel cryopreservation procedure. Comparison was made considering the results of both sperm motility and morphometry parameters. Moreover, because sperm cryopreservation in fish often depends on the use of permeating cryoprotectants bearing chemically reactive methyl group, we wanted to decipher whether eel sperm DNA methylation pattern was affected by the different protocols.
Material and methods
Male eels were matured with hCG weekly injections. After 10 weeks of treatment sperm samples were extracted and maintained at 4 ºC until use. Sperm motility was determined after activation with sea water and only those samples having >68% of motile cells were selected for the experiment. Four pooled samples, containing sperm from 3 different males were frozen (5 straws per sample) using both Asturiano et al. (2003) and Müller et al. (2004) protocols. In general, sperm was diluted with a mixture of extender and cryoprotectant, loaded into 0.5 or 0.25 ml straws and frozen in the vapor of liquid nitrogen. Straws were thawed by immersion into water bath for a given period of time.
Fresh and post-thawed samples were evaluated per triplicate (using three straws per pool) by sperm motility (CASA) and morphometry (ASMA) parameters. Two frozen straws per pool were sent to INRA´s lab, where the degree of DNA methylation and histone modification were evaluated in fresh and frozen-thawed samples using the restriction enzyme assay and assessing methylation by image analysis and by LUMA.
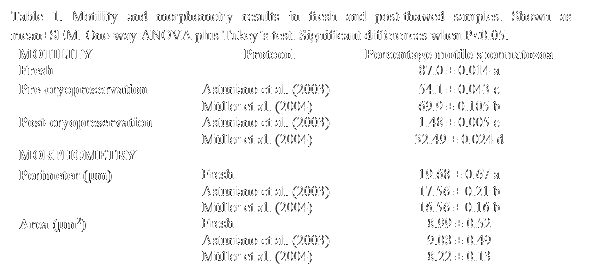
Results
Both methods caused a reduction on the percentage of motile cells (Table 1) that was measured after diluting the sperm in the different freezing media (and induced lower percentages with the "Spanish method"), and especially post-thawing, when the "Hungarian method" caused an evident higher percentage of motile cells. The ASMA morphometry analyses showed that both protocols induced a significant reduction of the perimeter of the spermatozoa´s head in comparison with fresh samples, but without significant differences between them. No differences were found in the cells head area. However, the "Hungarian method" caused slightly smaller values of both post-thawing perimeter and area, suggesting a higher dehydration caused by methanol in comparison with DMSO used in the "Spanish method".
The epigenetics evaluation found a general high grade of methylation, but no differences between samples cryopreserved with the two different protocols, neither the different pooled samples. However, the use of pooled samples could be masking the expected differences.
Discussion and conclusion
Preliminary results point the "Hungarian method" (Müller et al., 2012) as the most effective one for the cryopreservation of eel sperm, even when the reduction of morphometry parameters was slightly higher. However, new experiments must be carried out with individual sperm samples to avoid the compensatory effect of using pooled sperm samples on the damages affecting the epigenetic characteristics of sperm.
Acknowledgements
Funded by EU (PRO-EEL, grant agreement n°245257) and by the CRB-Anim project "Investissements d'Avenir" ANR-11-INBS-0003. M.M., E.K. and Á.H. had STSM grants from the COST Office (COST Action FA1205: AQUAGAMETE).
References
Asturiano, J.F., Pérez, L., Marco-Jiménez, F., Olivares, L., Vicente, J.S., Jover, M., 2003. Media and methods for the cryopreservation of European eel (Anguilla anguilla) sperm. Fish Physiology and Biochemistry, 28: 501-502.
Asturiano, J.F., Sørensen, S.R., Pérez, L., Lauesen, P., Tomkiewicz, J., 2013. Effect of preservation temperature and cryopreservation on European eel sperm fertilization capacity. First production of larvae using cryopreserved sperm. 4th International Workshop on Biology of Fish Gametes. Faro (Portugal). Book of abstracts, pp.: 154-155.
Müller, T., Urbányi, B., Váradi, B., Binder, T., Horn, P., Bercsényi, M., Horváth, Á., 2004. Cryopreservation of Sperm of Farmed European Eel Anguilla anguilla. Journal of the World Aquaculture Society, 35(2): 225-231.
Müller, T., Horváth, Á., Takahashi, E., Kolics, B., Bakos, K., Decsi, K., Kovács, B., Taller, J., Urbányi, B., Bercsényi, M., Horváth, L., Adachi, S., Arai, K., Yamaha, E., 2012. Artificial hybridization of Japanese and European eel (Anguilla japonica x A. anguilla) by using cryopreserved sperm from freshwater reared males. Aquaculture, 350-353: 130-133.
Peñaranda, D.S., Pérez, L., Gallego, V., Jover, M., Asturiano, J.F., 2009. Improvement of European eel sperm cryopreservation method by preventing spermatozoa movement activation caused by cryoprotectants. Cryobiology, 59(2): 119-126.
Szabó, G., Müller, T., Bercsényi, M., Urbányi, B., Kucska, B., Horváth, Á., 2005. Cryopreservation of European eel (Anguilla anguilla) sperm using different extenders and cryoprotectants. Acta Biologica Hungarica, 56(1-2): 173-175.