PHOTOBACTERIOSIS OUTBREAK IN CAGE-REARED RACHYCENTRON CANADUM: PREDISPOSING CONDITIONS
Introduction
Photobacterium damselae subsp piscicida is a Gram-negative bacterium that causes pseudotuberculosis or pasteurellosis (Truper & Declari 1997). This is an important infection for cobia fish farming mainly due to the high mortality that in some cases reached 90-100% and its difficult control (Romalde 2002). This study reports the occurrence of a photobacteriosis outbreak in young cobias farmed in the northern coast of Sao Paulo state, Brazil, caused by P. damselae subsp piscicida and discusses their morphological and microbiological characteristics, predisposing conditions, probable pathogenesis and possible treatments.
Material and methods
Fish were obtained from a farm located on the northern coast of Sao Paulo state (23° 48' 54" S 45° 22' 14" O). Farmers reported fish with growth delay, lethargy and high mortality. The first sampling was performed in March when fish were approximately 45 days old. The process continued to develop and the mortality increased. The following samplings were performed in May, July and September 2011. Water temperature in the months of sampling varied between 19ºC to 28ºC. In all cases, fish showed clinical signs of disease. They were submitted for euthanasia by deep anesthesia in benzocaine solution. The weight was measured in grams (g) and the total length in centimeters (cm). After that, necropsy was performed. Samples were collected for histological, microbiological and genetic analysis. They were performed antibiogram test, quantification of total lipids and fatty acid profile.
Results
The fish farm studied started with approximately 20 000 fries in November 2010, and after the infectious outbreak in September 2011, there were only 500 fish left. Weight and total length did not change between May and September 2011.
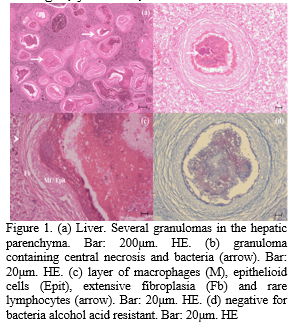
The fish presented ulcers around the dorsal fin with bone exposure, depigmentation, erosion of tegument, and loss of muscle mass. The coelomic cavity presented accumulation of a transparent liquid. Liver were enlarged, yellowish, soft whitish firm tubercles measuring from 0.5 to 2.0 mm of diameter distributed diffusely or multifocally. On the histophatological examination, it was observed that hepatocytes presented large vacuoles with nuclei displaced to the cytoplasm's periphery and lesions with necrotic centers containing bacterial accumulations limited by macrophages, few lymphocytes, and extensive fibroplasia (Figure 1).
In the microbiological analysis, it was observed a gram-negative, non-motile, oxidase positive and catalase positive bacteria. Alcohol acid resistant bacteria were negative for Ziehl-
Neelsen staining. The results of API 20E kit
system were compatible to the description of
Photobacterium damselae. PCR technique
using two pairs of primers oriented towards
the internal regions of 16S rRNA was used to confirm Photobacterium damselae subsp. piscicida. The bacterium was susceptible to two antibiotics: chloramphenicol and enrofloxacin. Total lipids and fatty acid profile of cobias' feed were presented previously in Shimada et al. (2014). It stands the imbalance in the proportion of fatty acids. Mainly, the high levels of oleic acid and the low levels of docosahexaenoic acid.
Discussion and Conclusion
The enlarged yellowish livers, that in the microscopically exam were compatible with steatosis, indicate lipid accumulation in the cell cytoplasm which is generally a reflex of nutritional or metabolic disorders (MacGavin and Zachary, 2006). In our study, livers showed severe and diffuse steatosis involving the entire liver parenchyma which may cause liver failure. This mechanism associated with the unbalanced feed may be related to the development disorder of the cobias.
According to Table 1, there was no difference in fish weight and size. It is noteworthy that muscles were largely consumed. Cobia in normal conditions can earn up to 7 kg in 16 months with feed conversion of 1.1:1 (Stevens 2004). This condition may also relate to the severe steatosis described in these fish.
The lesions observed are common in infections by P. damselae subsp. piscicida and damselae, but the former also presents whitish tubercles in the liver, spleen and kidneys (Bakopoulos et al. 2003). These tubercles were observed in this study, mainly in the liver and kidney. These are consistent with the chronic form of the disease caused by P. damselae subsp. piscicida (Liu et al. 2003).
In the tubercles described in this study, the identification of the etiological agent was performed according the characteristics of lesions and the phenotype of the microorganism relating to its physiological and biochemical particularities in API 20E kit to differentiate between the two subspecies of P. damselae (Romalde 2002). However, the genotypic analysis was essential to confirm the infection by P. damselae subsp. piscicida (Rajan et al. 2003). We discarded mycobacteriosis and nocardiosis due to bacteria culture characteristics, biochemical identification, negative Ziehl-Neelsen staining, and molecular analysis. The antibiogram showed that this bacterium presented sensitivity only for chloramphenicol and enrofloxacin.
In this study, the fish presented delayed growth, nutritional imbalance of fatty acids, moderate to severe hepatic steatosis and infection by P. damselae subsp. piscicida, an opportunistic bacterium. This presented sensitivity only for chloramphenicol and enrofloxacin, which can be used for treatment. This study suggests that steatosis may be a predisposing factor to photobacteriosis.
References
Bakopoulos, V., Volpatti, D., Gusmani, L., Adams, A., Dimitriadis, G. 2003. Vaccination trials of sea bass, Dicentrarchus labrax (L.), against Photobacterium damselae subsp. piscicida, using novel vaccine mixtures. Journal of Fish Disease 28: 77-90.
Liu, P., Lin, J., Lee, K. 2003. Virulence of Photobacterium damselae subsp. piscicida in cultured cobia Rachycentron canadum. Journal of Basic Microbiology 43: 499-507.
MacGavin, M.D., Zachary, J.F. 2006. Pathologic Basis of Veterinary Disease. 4 ed. Mosby.
Rajan, P.R., Lin, J.H.Y., Ro, M.S., Yang, H.L. 2003. Simple and rapid detection of Photobacterium damselae ssp. piscicida by a PCR technique and plating method. Journal Applied Microbiology 95: 1375-1380.
Romalde, J.L. 2002. Photobacterium damselae subsp. piscicida: an integrated view of a bacterial fish pathogen. International Microbiology 5: 3-9.
Shimada, M.T., Claudiano, G.S., Engracia Filho, J.R., Yunis, J., Moraes, F.R., Moreira, R.G., Moraes, J.R.E. 2014. Hepatic Steatosis in Cage-Reared Young Cobia, Rachycentron canadum (Linnaeus, 1766), in Brazil. Journal of Veterinary Science & Medical Diagnosis 3:2.
Spisni, E., Tugnoli, M., Ponticelli, A., Mordenti, T., Tomasi, V. 1998. Hepatic steatosis in artificially fed marine teleosts. Journal Fish Diseases 21: 177-184.
Stevens, O. 2004. ACFK: Cobia Fingerling Update. Global Aquaculture Advocate. February, p. 46-47.
Truper, H.G., Declari, L. 1997. Taxonomic note: necessary correction of epithets formed as substantives (nouns) "in apposition". International journal of systematic bacteriology 47: 908-909.